Increasing the pool of donor organs with new means of pre-transplant assessment and conditioning.
Ex vivo lung perfusion (EVLP) is a process of evaluating and preparing donor lungs outside the body prior to transplant surgery. The lungs are warmed to normal body temperature, flushed of donor blood, inflammatory cells and potentially harmful biologic factors, and treated with antibiotics and anti-inflammatory agents.
Frank D'Ovidio, MD, PhD, Surgical Director of the Lung Transplant Program at NewYork-Presbyterian/Columbia, is very pleased with the progress of the NOVEL trial of Ex Vivo Lung Transplantation, which had begun shortly before it was highlighted in the Summer 2012 issue of Healthpoints. "Ex vivo perfusion is a valuable resource to assess organs and potentially improve some that would have been turned down because of quality."
Already approved and in use in Europe and in Canada, this process of evaluation and reconditioning is of great interest because it can increase the pool of available donorlungs that are suitable for transplantation. Because so many donor lungs are damaged at the time of death, only 20-30% of donated lungs are usable. Since the initiation of the FDA "NOVEL" trial, the lung transplant team at NYP/Columbia has evaluated twelve sets of lungs and has been able to perform eight transplants that would not have been possible without EVLP.
All the NYP/Columbia EVLP transplant recipients are doing well and their outcomes are equivalent to those of patients who received lungs that had not been subject to EVLP, confirming what has already been established about the safety of this process.
Lung Transplant Facts
- Once you get into the NYP/CUMC transplant program, you'll be placed on the Organ Procurement and Transplantation Network's (OPTN's) national waiting list. OPTN matches donor lungs to recipients based on need, severity of disease, and whether the transplant will improve a recipient's chance of survival.
- Approximately 1700 people are on the waitlist for lung transplantation in the U.S.
- Each year, about 1400 lung transplants are performed.
- Since the organ allocation system was revised in 2005, the average wait time for lung transplantation dropped from about 2 years to 3-4 months, an improvement which has reduced the risk of dying while on the organ waitlist.
Organ Care Recovery Unit
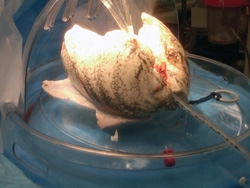 Not only may EVLP increase the pool of donor lungs suitable for transplantation, but it holds exciting potential to open other possibilities in the field of organ transplantation. Dr. D'Ovidio and colleagues are developing plans to establish an Organ Care Recovery Unit, which would allow intensive care of lungs that were inappropriate for immediate transplantation. "We would assess these organs toward the goal of reconditioning them for transplant if appropriate," he explains.
Not only may EVLP increase the pool of donor lungs suitable for transplantation, but it holds exciting potential to open other possibilities in the field of organ transplantation. Dr. D'Ovidio and colleagues are developing plans to establish an Organ Care Recovery Unit, which would allow intensive care of lungs that were inappropriate for immediate transplantation. "We would assess these organs toward the goal of reconditioning them for transplant if appropriate," he explains.
The Organ Care Recovery Unit would also apply the ex vivo assessment and perfusion technique to organs other than lungs, potentially offering the same benefit of increasing access to transplantation for patients in need of a kidney, liver, heart, or pancreas. The methodology is currently being tested with donor livers and kidneys.
Moreover, the success of EVLP has opened a new channel of research into tailoring treatment of donor lungs to benefit the recipient. Dr. D'Ovidio anticipates that in the coming years, researchers will administer cell therapies, gene therapies, and pharmacologic agents during EVLP to determine whether such treatments will improve early and long-term outcomes of lung transplantation. "As research develops, we may also be able to use more specific therapies to personalize the treatment based on the understanding of the genetic background of the lungs," Dr. D'Ovidio explains. Related research is currently underway; stay tuned for more on this topic in future issues.