Guide to Breast Cancer
Reviewed by the physicians of Columbia’s Surgical Breast Team, December 2023
Anatomy | How Does Breast Cancer Spread | Risk Factors | Symptoms | Diagnosis | Staging | Treatments | Next Steps
Key Info
- One in eight women in the U.S. will be diagnosed with breast cancer in their lifetime and over 220,000 women are diagnosed each year.
- Breast cancer is the most commonly diagnosed cancer in women. It is rare in men, with approximately 2,150 cases diagnosed each year.
- Mammography is recommended for most suspicious breast lumps but a biopsy is often needed to confirm the diagnosis.
What is breast cancer?
Like cancer in other parts of the body, breast cancer involves the growth and multiplication of abnormal cells.
When the normal process of cell growth is disrupted, new cells begin to form when the body doesn’t need them and old or damaged cells fail to die when they should. These abnormal cells often form a mass of tissue called a lump or tumor. These growths can be benign (not dangerous to a patient’s health) or malignant (in need of immediate treatment).
When malignant cells form in the tissues of the breast, they can invade the surrounding tissue or spread to other areas of the body. With early detection and treatment, however, most breast cancer patients continue to lead a normal life.
Breast cancers may be either hormone positive or hormone negative, meaning that some tumors are fueled by the body’s production of estrogen or progesterone (hormone positive), while others are not.
Anatomy
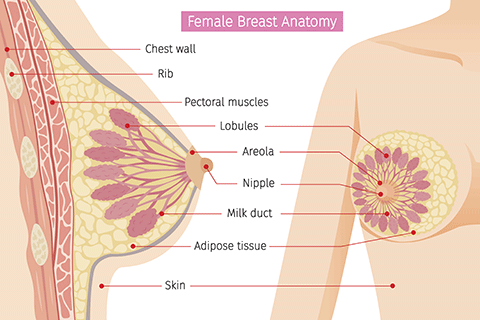 The breast is mostly made up of fatty tissue. Within this tissue is a network of lobes, which are made up of tiny, tube-like structures called lobules that contain milk glands. Tiny ducts connecting the glands, lobules, and lobes, carry the milk to the nipple.
The breast is mostly made up of fatty tissue. Within this tissue is a network of lobes, which are made up of tiny, tube-like structures called lobules that contain milk glands. Tiny ducts connecting the glands, lobules, and lobes, carry the milk to the nipple.
Blood and lymph vessels also run throughout the breast; blood nourishes the cells, and the lymph system drains bodily waste products. The lymph vessels connect to lymph nodes, the tiny, bean-shaped organs that help fight infection.
Most commonly, breast cancer begins in the ducts that carry milk to the nipple (called ductal carcinomas). Almost 75 percent of all breast cancers originate here.
Less commonly, lobular carcinomas are those that develop in the cells lining the lobules that produce milk. When a ductal or lobular carcinoma does not spread beyond the original duct or lobe, it is termed in situ; cancers that spread to other tissues are called invasive cancers.
More uncommon are breast sarcomas, which originate in the bone, muscle, fat, or connective tissue.
Other, less common types of breast cancer include medullary, mucinous, tubular, metaplastic and papillary breast cancer.
Inflammatory breast cancer is a faster-growing type of cancer accounting for about 1% to 5% of all breast cancers.
How Does Breast Cancer Spread
Breast cancer spreads when the cancer cells show up in other parts of the body or when breast cancer cells travel to other organs through the blood vessels and/or lymph vessels. This is called metastasis.
Breast cancer most often spreads to the lymph nodes under the arm (axillary nodes), neck (cervical nodes), under the chest bones (internal mammary nodes), or just above the collar bone (supraclavicular nodes).
When it spreads throughout the system, it can appear in the bones, lungs, and liver. Less often, breast cancer may affect the brain.
Breast cancer can also recur (come back after treatment), appearing locally in the skin, in the same breast (if it was not removed as part of treatment), in other tissues of the chest, or elsewhere in the body.
Risk Factors for Breast Cancer
One in eight women in the U.S. will be diagnosed with breast cancer in their lifetime and over 220,000 women are diagnosed each year.
In recent years, there has been a gradual reduction in breast cancer incidence rates among women aged 50 or older. Death rates from breast cancer have also been declining since about 1990, in part due to better screening and early detection, and the development of better treatment options. There are over 2 million breast cancer survivors today.
Risk factors for developing breast cancer include:
Gender
Breast cancer is the most commonly diagnosed cancer in women. It is rare in men, with approximately 2,150 cases diagnosed each year. The reason is probably that men have less of the “female” hormones estrogen and progesterone, which can promote the growth of breast cancer cells.
Age
Roughly one out of every eight invasive breast cancers are discovered in women younger than 45. In contrast, two out of three invasive breast cancers are found in women 55 or older.
Genetic factors
Between 5 percent and 10 percent of breast cancers are thought to be hereditary, meaning that they result directly from a defect or mutation in the genes. This defect is hereditary, meaning that it can be passed on by a parent.
The BRCA1 and BRCA2 genes are the most common cause of hereditary breast cancer. Under normal circumstances, these genes help prevent cancer. Their job is to make proteins that keep the cells from growing abnormally, but when these genes are damaged, they do not function properly.
Women carrying inherited mutations in the BRCA1 and BRCA2 genes have an extremely high lifetime risk for developing breast and/or ovarian cancer. Moreover, breast cancer occurs at an earlier age in these patients. More pathological features have also been reported in breast cancers occurring in BRCA1 mutation carriers. Read more here about our genetic counseling program.
Family history
A first-degree relative (mother, sister, or daughter) with breast cancer roughly doubles a woman's risk. Two first-degree relatives with breast cancer increase a woman’s risk about three times.
Women with a father or brother diagnosed with breast cancer, also have an increased risk. However, it is important to note that 85% of all women diagnosed with breast cancer do not have a family history of this disease.
Personal history
A woman with cancer in one breast has a three to four times greater chance of developing a new cancer in the other breast or in another part of the same breast. This is considered an independent growth—not a recurrence (a return) of the first cancer.
Race
White women have a slightly higher chance of developing breast cancer than African-American women. Yet in women under 45, breast cancer is more common in African-American women.
Asian, Hispanic, and Native-American women have a lower risk of developing and dying from breast cancer.
Dense breast tissue
Women with dense breasts have a higher risk of breast cancer. Unfortunately, dense breast tissue can also make mammograms less accurate.
A number of factors affect breast density, such as age, menopausal status, the use of hormone therapy during menopause, pregnancy, and genetics. A “dense” breast (as seen on a mammogram) has more glandular and fibrous tissue and less fatty tissue.
Read more here about dense breast tissue
Benign breast conditions
- Non-proliferative lesions generally don’t affect breast cancer risk, or may only increase it very little. These include simple cysts, what used to be called “fibrocystic changes”, certain benign tumors and calcifications, mild hyperplasia, a single papilloma, adenosis (non-sclerosing), ductal ectasia, periductal fibrosis, squamous and apocrine metaplasia and fat necrosis.
- Proliferative lesions without atypia are related to excessive growth of cells in the ducts or lobules of the breast tissue and appear to raise a woman's risk of breast cancer slightly (one-and-a-half to two times normal). These conditions include fibroadenoma, sclerosing adenosis, several papillomas, and radial scars.
- Proliferative lesions with atypia involve an overgrowth of cells in the ducts or lobules of the breast tissue, with some of the cells no longer appearing normal. These conditions elevate breast cancer risk to three-and-half to five times higher than normal.
Read more here about non-cancerous breast disease
Lobular carcinoma in situ (LCIS)
In this condition, abnormal cells appear in the lobules of the milk-producing glands of the breast, but do not grow through the wall of the lobules. This diagnosis is linked to a seven to eleven-fold increased risk for developing invasive cancer (in either breast).
More years menstruating
Women who started menstruating before age 12 and/or started menopause after age 55, have a slightly higher risk of breast cancer. This may be due to the body’s longer production of the hormones estrogen and progesterone.
Not having children
Women who haven’t had children or who gave birth after age 30 have a slightly higher risk for developing breast cancer, while those who’ve had several pregnancies and started their families at a younger age, have reduced risk.
Oral contraceptives
Women using oral contraceptives (birth control pills) have a slightly higher risk of breast cancer. This risk begins to drop once the pills are stopped, and to go back to normal 10 years after a woman has stopped taking these pills.
Post-menopausal Hormone Therapy (PHT)
PHT, also known as hormone replacement therapy (HRT), and menopausal hormone therapy (MHT), increases breast cancer risk.
Combined HT (the use of estrogen and progesterone) can elevate risk in as little as two years. It also increases the likelihood that the cancer may be found at a more advanced stage. However, a woman's breast cancer risk seems to return to that of the general population within five years of stopping combined treatment.
Alcohol Use
One alcoholic drink a day carries a very small increase in risk. Yet women who have two to five drinks daily have about one-and-a-half times the risk of developing breast cancer than those who don’t drink at all.
Obesity/Overweight
Breast cancer risk is greater for women who gained a significant amount of weight as an adult but the risk may not be increased at all for women who have been overweight since childhood. Also, excess fat around the waist may have more of an impact on risk than fat around the thighs or hips.
Previous chest radiation
Women who in their childhood or adolescence, received radiation therapy to the chest to treat another cancer have a significantly increased risk for breast cancer. The chance of developing breast cancer from chest radiation is greater if the radiation was given when the patient’s breasts were still developing. Radiation treatment after age 40 does not seem to increase breast cancer risk. If the patient was treated with chemotherapy as well, this may have stopped ovarian hormone production long enough to lower risk.
Diethylstilbestrol (DES) exposure
Until the 1960s the drug diethylstilbestrol (DES) was prescribed for pregnant women to lessen the chance of miscarriage (losing the baby). These women have a slightly increased risk of developing breast cancer. Women whose mothers took DES during pregnancy may have a slightly higher risk of breast cancer.
Symptoms of Breast Cancer
A simple self-exam helps patients to keep track of any changes in the breast. Among the things to look for—though not necessarily signs of cancer—are:
- Nipple tenderness
- Any nipple discharge
- A lump or thickening in or near the breast or in the underarm area
- A lump in the breast (not all lumps are cancerous but should be evaluated by a physician)
- An unusual change in the size or shape of the breast
- An usual swelling of the breast (especially if on one side only)
- Shrinkage of the breast (especially if on one side only)
- A new asymmetry of the breasts
- A nipple that has turned slightly inward or become inverted
- Scaly, red or swollen skin on the breast, areola, or nipple
- Ridges, dimpling or pitting in the breast, resembling the skin of an orange
Diagnosis of Breast Cancer
Breast Imaging technologies allow physicians to detect cancer at an early stage, when patients receive the optimal effects from treatment.
Mammography
For over thirty years, mammography—an x-ray image involving the compression of the breast—has been an important tool for early diagnosis and it is recommended that women over age 40 have a screening mammogram each year.
Mammography is recommended for most suspicious breast lumps but a biopsy is often needed to confirm the diagnosis.
The sensitivity of mammography decreases as the density of the breast increases. However, one of its great benefits is to detect microcalcifications—tiny spots of calcium in the breast. While often benign, these sometimes indicate the presence of ductal carcinoma in situ (DCIS), a very early stage of breast cancer. Microcalcifications are not detected by MRI or by ultrasound.
Read more here about the usefulness of mammography
Learn more about breast cancer screening and prevention
Ultrasound
This approach uses sound waves to take images of the breast. The skin is usually lubricated with gel, then a small, microphone-like instrument called a transducer is placed on the breast. The transducer emits sound waves and picks up the echoes as they bounce off body tissues. These echoes then appear as a black and white image on a computer screen. This test is painless and does not expose you to radiation.
Breast ultrasound may be a helpful addition to mammography when screening women with dense breast tissue. It may also be useful for evaluating some breast masses and to determine if a suspicious area is a cyst (fluid-filled sac). In addition, ultrasound can help doctors guide a biopsy needle into some breast lesions.
Magnetic Resonance Imaging (MRI)
For patients at high-risk, the most appropriate screening tool is an MRI, using magnetic fields and radio waves to create a detailed image of the breast. In the “high-risk” category are women who have at least a 20-25 percent chance of developing breast cancer, including those with
- Mutations of BRCA1/2 genes
- A family history of breast cancer (mother, daughter, sister)
- Previous treatment for Hodgkins disease including radiation to the thorax
- Previous diagnosis of breast cancer
In some cases, women with high-risk lesions, atypia, or lobular carcinoma in situ are considered candidates for an MRI.
Biopsy
When a lump is found, a needle biopsy can be performed in the physician’s office. A local anesthetic is injected into the breast, and light suction is applied through a hollow needle inserted into the lump to remove a sample of tissue or fluid. Disappearance of the lump after fluid is withdrawn usually indicates a benign cyst. Nevertheless, the fluid is sent to a laboratory for analysis. If no abnormal cells are found, no further tests are required.
In some cases, a surgical biopsy may be necessary. This procedure entails removing the lump and small amounts of surrounding tissues for laboratory analysis. A surgical biopsy usually is done in a hospital (often as out-patient surgery) using either local or general anesthesia. About 80 percent of these biopsies show no cancerous cells.
Additional Tests
When a tumor is found, it is tested to determine whether it is hormone dependent (estrogen- or progesterone-receptor positive). Additional tests are sometimes performed to measure the rate at which tumor cells are dividing (DNA flow cytometry) and to detect presence of abnormal genes such as one called Her 2/Neu.
Staging of Breast Cancer
Once the tumor has been identified, breast cancer will be classified according to its stage of growth. This will help determine the best possible treatment.
- Stage 0 (early stage): Includes findings of ductal carcinoma in situ (DCIS) in which the tumor has not invaded outside of the breast ducts.
- Stage I (early stage): Tumor no more than 2 cm across, with no spread to lymph nodes.
- Stage II (early stage): Tumor 2 to 5 cm across, and cancer has spread to underarm lymph nodes.
- Stage IIA: Tumor less than 2 cm with lymph node involvement, or cancer between 2 and 5 cm with no lymph node involvement.
- Stage IIB: Tumor larger than 2 cm with lymph node involvement.
- Stage III: Also called locally advanced cancer, tumor larger than 5 cm, with extensive cancer in underarm lymph nodes, or that has spread to other lymph node areas or tissues near the breast (such as above or below the clavicle).
- Stage IIIA: Tumor larger than 5 cm with involvement of underarm lymph nodes. Any tumors fitting the description above.
- Stage IIIB: As in IIIA, and associated with chest wall or skin involvement, or with spread to lymph node above the clavicle.
- Stage IV: Cancer has spread to another organ in the body (also called metastatic cancer).
How is Breast Cancer Treated?
Chemotherapy
Chemotherapy drugs are given either before the surgery to shrink the tumor (neo-adjuvant therapy), or after the surgery, to keep the cancer from growing back (adjuvant therapy.)
Chemoprevention/Hormonal Therapy
This approach involves the use of systemic agents to lower a patient’s risk of developing cancer by blocking the body’s natural production of hormones. Tamoxifen is approved for use in prevention, but is associated with serious side effects (including hot flashes, increased risk of uterine cancer, and blood clots). A similar drug, raloxifene, has fewer side effects and was recently approved by the FDA to reduce risk of breast cancer.
Targeted Therapies
This type of treatment attacks specific types of cancer cells with less harm to normal cells. Some types of targeted therapies help the immune system kill cancer cells or deliver toxic substances directly to cancer cells and kill them. These are called immunotherapy drugs.
Other targeted therapies block the action of certain proteins involved in the growth and spread of cancer cells. These are called monoclonal antibodies.
Targeted therapy may have fewer side effects than other types of cancer treatment.
Surgery for Breast Cancer
Operations to treat breast cancer may include the following:
Breast-conserving surgery: This approach removes the cancer while leaving as much breast tissue as possible intact. This includes lumpectomy (removal of the lump with some surrounding tissue at the margin of the tumor), quadrantectomy (removal of one quarter, or quadrant, of the breast), and segmental mastectomy (removal of the cancer as well as some of the breast tissue around the tumor and the lining over the chest muscles below the tumor).
Modified radical mastectomy: In this operation, the surgeon removes the breast, most or all of the lymph nodes under the arm, and the lining over the chest muscles. In some cases, part of the chest wall muscles will be removed as well. This may be followed by reconstructive surgery to reshape the breast.
Total mastectomy: This surgery, also called simple mastectomy, removes the entire breast.
Lymph node removal: Following surgery, the patient’s lymph nodes may be removed and a sample of tissue taken, to check if the cancer has spread. In what is called a regional lymph node dissection, some of the lymph nodes in the tumor area are removed. In a radical lymph node dissection, most or all of the lymph nodes in the tumor area are removed. This procedure is also called a lymphadenectomy. A common side-effect of breast cancer surgery is lymphedema, swelling in the arms, hands, fingers, shoulders or chest, caused by fluid retention in the body. Columbia surgeons have developed innovative protocols to prevent lymphedema following surgery for breast cancer.
Oncoplastic Breast Surgery: Surgeons at the Columbia’s Clinical Breast Cancer Program are fully versed in Oncoplastic Surgical techniques, which provide excellent cosmetic results while maintaining sound oncologic principles (effective removal of cancerous tissue) following major resection of breast cancers.
Next Steps
If you or a loved one have been diagnosed with breast cancer, we’re here for you. Call us now at (212) 305-9676 or fill out our online appointment request form.
Related
- Non-Cancerous Breast Disease
- BRCA Genes and Breast Cancer
- Breast Screening and Prevention
- Breast Surgeries & Procedures
- Oncoplastic Breast Surgery
- Intraoperative Radiation Therapy for Breast Cancer
- Reconstructive Surgery
